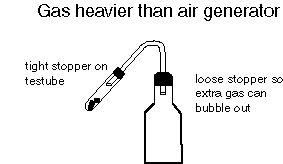Molecular Weight of a Gas
Purpose:
You will be collecting one of the following gases by displacement of either air or water.
Materials:
- Get the chemicals needed for the gas you are going to produce.
- Oxygen gas--4 grams of 33% manganese dioxide and 67% potassium chlorate by mass.
- Hydrogen gas--zinc metal and sulfuric acid.
- Carbon dioxide gas--20 grams of calcium carbonate and hydrochloric acid.
- Testube with a one hole stopper and glass rod bent to shape.
- Collecting bottle with a stopper.
- barometer
- balance or scale
Materials:
- Get the chemicals needed for the gas you are going to produce.
- Oxygen gas--4 grams of 33% manganese dioxide and 67% potassium chlorate by mass.
- Hydrogen gas--zinc metal and sulfuric acid.
- Carbon dioxide gas--20 grams of calcium carbonate and hydrochloric acid.
- Testube with a one hole stopper and glass rod bent to shape.
- Collecting bottle with a stopper.
- barometer
- balance or scale
- Oxygen gas--4 grams of 33% manganese dioxide and 67% potassium chlorate by mass.
- Hydrogen gas--zinc metal and sulfuric acid.
- Carbon dioxide gas--20 grams of calcium carbonate and hydrochloric acid.
Procedure:
- Take your stoppered bottle and immediately weigh it with air.
- Record the barometric pressure and temperature of the air.
- Fill the bottle with water up to the bottom of the stopper and then measure the amount of water (this is the amount of gas the bottle holds).
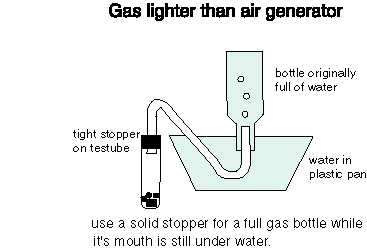
- See the diagram below for connecting up the testube to the bottle.
- Mix the chemicals in the testube and immediately stopper securely.
- Let some of the gas produced run out of the tubing before placing into the collecting bottle.
- Fill your bottle with gas and then stopper.
- If the bottle and stopper are wet, dry them carefully so the stopper does not come out.
Data Table:
mass of bottle and stopper with air_________________
mass of bottle and stopper with gas_________________
temperature of gas_____________
barometer reading______________
volume of bottle to stopper (water vol.)____________
mass of assembly_______________
volume of gas corrected to STP_______________
molecular weight of gas (experimental)_______________
molecular weight of gas (accepted)___________________
percent error_______________