Titration Lab
You will be making up a 200 ml. standardized basic solution of sodium hydroxide (NaOH) of a recommended concentration of 0.5 to 3 M. Have your math checked before weighing out the chemical.
Put the solid into a 100 ml. graduated cylinder and add water to the top mark. Dissolve all the sodium hydroxide before continuing. Pour the solution into a clean beaker. Add another 100 ml. of distilled water for a total volume of 200 milliliters.
Measure out 10 ml. of the unknown acid into a clean beaker. Add a few drops of indicator (litmus or universal). Place the beaker on white paper under the buret containing your know base. Titrate to the end point (color change).
Do all the acids indicated and find their concentration.
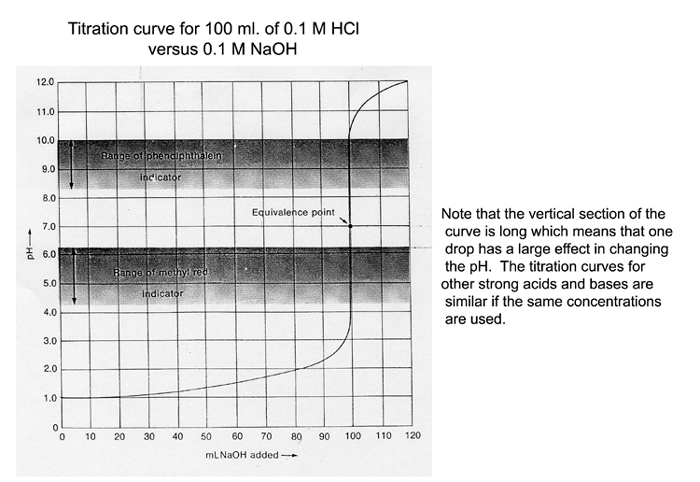
The conclusion should contain the following:
A. How can acids change concentration over time?
B. When would phenolphthalein be used?
C. What is the value of using the universal
indicator instead of litmus?
D. Show all reactions and indicate the conjugate
acid-base pairs.
E. Tell what kind of salt was formed such as
acid salt, etc.
Back to top



